Arkansas Plant Disease Clinic
Do you have a plant disease that you need to have identified? Visit this site to learn how to take a sample and where to send your sample.
See how the Plant Health Clinic can be of benefit
Trees, like any other living thing, are susceptible to diseases. Some of the diseases encountered in trees are merely unsightly whereas others can reduce productivity or kill the tree.
When the defenses against disease are compromised, the disease can gain a foothold in the tree resulting in infection and sometimes in tree death.
Many environmental factors can cause a tree to be stressed.
Good tree management reduces drought stress and mechanical damage and thus reduces the likelihood of disease problems with trees.
Do you have a plant disease that you need to have identified? Visit this site to learn how to take a sample and where to send your sample.
See how the Plant Health Clinic can be of benefitMost of the common foliar diseases are fungal. Some of the diseases can be lethal to trees, while most are merely unsightly. Treatment, or lack of it, must be based on the threat presented by the disease compared to the cost of treatment; therefore, careful attention must be paid to disease identification. Be aware that chemical injury and insects can mimic some of these diseases. Prevalence of foliar disease often is influenced by weather, so little can be done to prevent or treat the disease. The most practical control for some of these diseases is to simply rake and destroy leaves, the source of next year's infection, in the fall.
Needle rusts (right) grow on the surface of the needle and cause no real harm.

Needle casts (left) primarily grow inside the needle and often cause it to be shed.
Needle Blights

Needle blights (right) primarily grow inside the needle and cause part of it to die.
Treatment of these needle diseases must be based on the particular disease, the size of the tree involved, and the setting. Most of the needle diseases are merely unsightly and pose no long-term threat to the tree’s survival. In those cases, no treatment is necessary unless the disease reduces the trees immediate merchantability. Some trees are large enough that tree size alone limits treatment options. Ornamental trees and Christmas trees have a much greater value than forest trees and may warrant a greater investment in disease treatment.
See Extension fact sheet FSA5022, Pine Needle Diseases in Arkansas, for more information.
Hardwood Leaf Diseases
There are many foliar diseases of hardwood trees, but chemical injury and insects can mimic some of these diseases. Foliar disease often is a function of weather and little can be done to prevent or treat the disease. The most practical control for some of these diseases is to simply rake and destroy leaves, the source of next year's infection, in the fall.

Anthracnose is a fungal disease that affects primarily the leaves but also sometimes the stems of hardwood trees. Severe damage can occur but there is very little that can be done to minimize the damage. The most common symptom of these diseases is dead areas or blotches on the leaves. Because of the scorched appearance of the leaves, the diseases are sometimes called leaf blight. Sycamore, dogwood, maple, hickory, walnut, birch, oak, and ash are susceptible.
These University of Arkansas fact sheets FSA7533, Anthracnose Diseases of Common Landscape Trees, and FSA7564, Anthracnose Diseases of Dogwood, along with U.S. Forest Service publication, Anthracnose Diseases of Eastern Hardwoods, provide more information about anthracnose diseases.

Rusts are fungal diseases that affect leaves of several hardwood species. Leaf rust is a common problem, but rarely is serious. It is characterized by yellow spots on the upper leaf surface. Close examination reveals small yellow-orange bumps filled with powdery spores on the leaves. As with leaf spot, rust infestations will become apparent in mid to late August. The biggest effect on trees is early defoliation that would affect growth. Poplars and cottonwoods are susceptible to Melampsora rust and improved varieties have been bred for resistance. Most rust fungi have alternative hosts during the life cycle. Rusts affect cottonwood, poplars, willows, oaks, ash, birch, maples, and plums.
This U.S. Forest Service publication, Melampsora Rusts: Common leaf rusts of poplars and willows, provides more information about Melampsora rusts.

Leaf spots on trees are generally not important in the scheme of things but can be unsightly. Several species of fungi may cause leaf spot. The most common are Septoria, Phyllosticta, Mycoshaerella, and Actinopelte. These spots are generally brown and often the fruiting bodies may be seen on the leaves. Some leaf spots are caused by a parasitic alga (Cephaleuros virescens). Cool wet springs are conducive to the formation of leaf spots. Septoria can also infect the stems of hybrid poplars forming a canker.
This University of Arkansas publication, FSA7553, Algal Leaf Spot of Magnolia, and this University of Illinois publication, Fungal Leaf Spot Diseases of Shade and Ornamental Trees in the Midwest, provide more information about leaf spot.

Tar spot on maple and sycamore is not actually "tar" on the leaves, but rather a fungal disease. These diseases seldom are detrimental to the overall health of infected trees. Tar spots may cause premature defoliation but are not known to kill trees. Tar spots on maples are caused by fungi in the genus Rhytisma. The most common species are Rhytisma acerinum and R. punctatum.
Symptoms first appear in late spring or early summer as infected leaves develop light green or yellow-green spots. During mid to late summer, black tar-like raised structures are formed on the upper surface of leaves within the yellow spots. The R. acerinum fungus causes spots that are 1/4 to 3/4 inch in diameter, R. punctatum causes spots that are smaller (about a 1/16 inch diameter). Spots caused by R. punctatum are sometimes called speckled tar spots. This Michigan State University publication, Maple Tar Spot, provides more information about tar spot.

Powdery mildew is one of the most common plant diseases and is easily recognized. White spots or patches with a talcum powder appearance characterize this disease. Powdery mildews are severe in warm, dry weather with succulent tissue being more susceptible to infection. Many plants have developed a resistance to powdery mildew, but chemical treatments are effective control when the disease crops up. Most powdery mildew fungi attacking trees belong to the genus Microsphaea. The fungus overwinters in the leaves and is moved to the host plants the following spring by insects, wind, or rain.
The University of Arkansas fact sheet FSA6113, Powdery Mildew of Landscape Ornamentals, and this University of Minnesota web page, Powdery Mildew, provides more information on the subject.

Oak leaf blister is caused by the fungus Taphrina caerulescens and occurs on most species of oaks. The spots on the leaves are ¼ to ½ inch diameter and turn a light green as the young leaves expand. The spots age and become covered with an off-white coating of fungal growth that later turns brown. Leaves will remain on the tree. Cool moist conditions during leaf development are required and the leaf can only be infected during leaf expansion. Dormant spores overwinter in the buds of susceptible trees. This disease is of little consequence to the tree.
This University of Arkansas publication, Leaf Blister of Oak, provides more information about leaf blister.
As with the foliar diseases, most of the stem disease are fungal. Because of the impact many of these diseases have on the vascular system of the tree, the phloem, many of these disease are lethal to parts of the tree crown or the entire tree. Infected branches may be removed to save the tree. See University of Arkansas fact sheet Ten Easy Ways to Kill a Tree (And How to Avoid Them) for pruning instructions. Unfortunately, trees with infected trunks can rarely be saved.
Most of the common diseases that infect the stems of pines are fungal. Fusiform rust and the very closely related eastern gall rust are the most common fungal diseases of pines in Arkansas. Trees that are young when infected with these rusts usually develop galls in the main stem and die. Older trees can also be infected and will develop galls in the branches; but the infections rarely migrate to the main stem, so they pose no real threat to the tree.
See Extension fact sheet FSA7543, Fusiform Rust in Arkansas, for more information.

Black knot is a disfiguring and potentially lethal disease of trees and shrubs in the genus Prunus. This genus includes stone fruits such as wild, fruit-bearing and ornamental plums and cherries. During the first year of infection, black knot-infected trees develop greenish-brown to brown swellings on affected branches and trunks. During the second year, these swellings enlarge into the ugly, black, erupting tumors (galls) characteristic of the disease. Older gall tissue (greater than two years old) often dies and then is colonized by fungi that give the gall a whitish or pinkish color. Severe black knot infections may cause general tree decline or death if galls girdle large limbs, or tree trunks. The fungus Apiosporina morbosa, causes black knot. Spores of the fungus are released from these galls and infect new branches in late spring/early summer during periods of wet weather and mild temperatures (55-75°F). Pruning is an effective control method for homeowners. Chemical control and pruning are used in orchards.
This publication from the U.S. Forest Service, Black Knot of Cherry and Plum, provides more information about black knot.

Localized areas of dead bark and underlying wood on twigs, branches and trunks are called cankers. Many fungi that cause cankers normally inhabit the surface of the tree, gain entrance through natural or man-made wounds, and only cause disease when the tree is under stress. However, some fungi aggressively attack trees and cause cankers.

Perennial cankers are seldom lethal to the tree but do weaken its structure and detract from its appearance. Wounds and branch stubs are invaded the fungi during the tree's dormant period. The host forms callus around the infection site during the growing season but the fungus invades more tissue the following dormant period. As this interaction continues, a target-spot canker forms. (Examples: Nectria canker, Eutypella canker) Diffuse cankers are elongate with little or no callus growth. The fungus invasion is so rapid, the tree tissue at the edge of the advancing fungus is killed rapidly. Branches or whole trees are girdled sometimes in a single season. These diffuse cankers often kill the tree. (Examples: Chestnut blight, Botryosphaeria canker, Phytophthora dieback, Cytospora canker, Hypoxylon canker). Cankers are generally started with some wound or cracking of the bark. The trees natural defense mechanism usually can resist most canker diseases, but stress, for example drought, will weaken the defense mechanism. Hypoxylon canker on oak is a good example of tree resistance until some stress causes the disease to be lethal. There is no good chemical control of cankers. Pruning of limbs are a great method to prevent spread but if a canker occurs on the trunk, it is better to remove the tree.
University of Arkansas fact sheet FSA7557, Hypoxylon Canker of Hardwood Shade Trees, provides more information about cankers. This publication from Iowa State University, Fungal Cankers of Trees, and this one from Cornell University, Cankers on Trees, also provide more information on the topic.

Nearly all decay is caused by fungi.
Mushrooms, sometimes called conks, are usually seen growing on the sides of trees. Trees are usually infected for many years before mushrooms are visible. Removing these conks will not cure the tree, since the fungus is living inside the tree.
Wood decay begins with any injury that breaks through the bark and exposes the wood. The injury may be the result of a string trimmer, animal damage, broken branch, etc. The damaged cells undergo chemical changes and become discolored. Many microorganisms land on the surface of the wound. The tree uses a natural process called compartmentalization as a defense against decay.
During compartmentalization, the tree produces three zones to help prevent invasion and spread of the fungus. These zones produce defense compounds, which try to wall off the fungus by plugging the vascular tissue and producing callus tissue around the edge of the wound. The effectiveness of these three zones depends upon the type of fungus and the host tree. Some fungi are specifically adapted to tolerate the chemical-response compounds of the tree and will survive in spite of the tree's defense mechanisms. Besides weakening trees, rot destroys wood quality in the tree.
This publication from the U.S. Forest Service, Wood Decay in Living and Dead Trees: A Pictorial Overview, and this one from the University of Kentucky, Wounds and Wood Decay of Trees, provide more information about tree decays.

Wilts are serious diseases in trees that quickly result in death. Most are caused by fungi that literally clog the vessel elements, thus blocking water flow to the tree crown, leading to a quick death. This is characterized by the scorched appearance of leaves that remain attached to the trees. Oak wilt, verticillium wilt, mimosa wilt and Dutch elm disease are good examples of wilts.
This U.S Forest Service web page, Oak Wilt, and these fact sheets, Verticillium Wilt of
Shade Trees from Perdue University and Dutch Elm Disease from the University of Kentucky provide additional information.

As with foliar and stem diseases, most of the diseases afflicting tree roots are fungal. These diseases usually are lethal to the tree in the long term because these diseases impact the ability of the tree to absorb water from the soil.
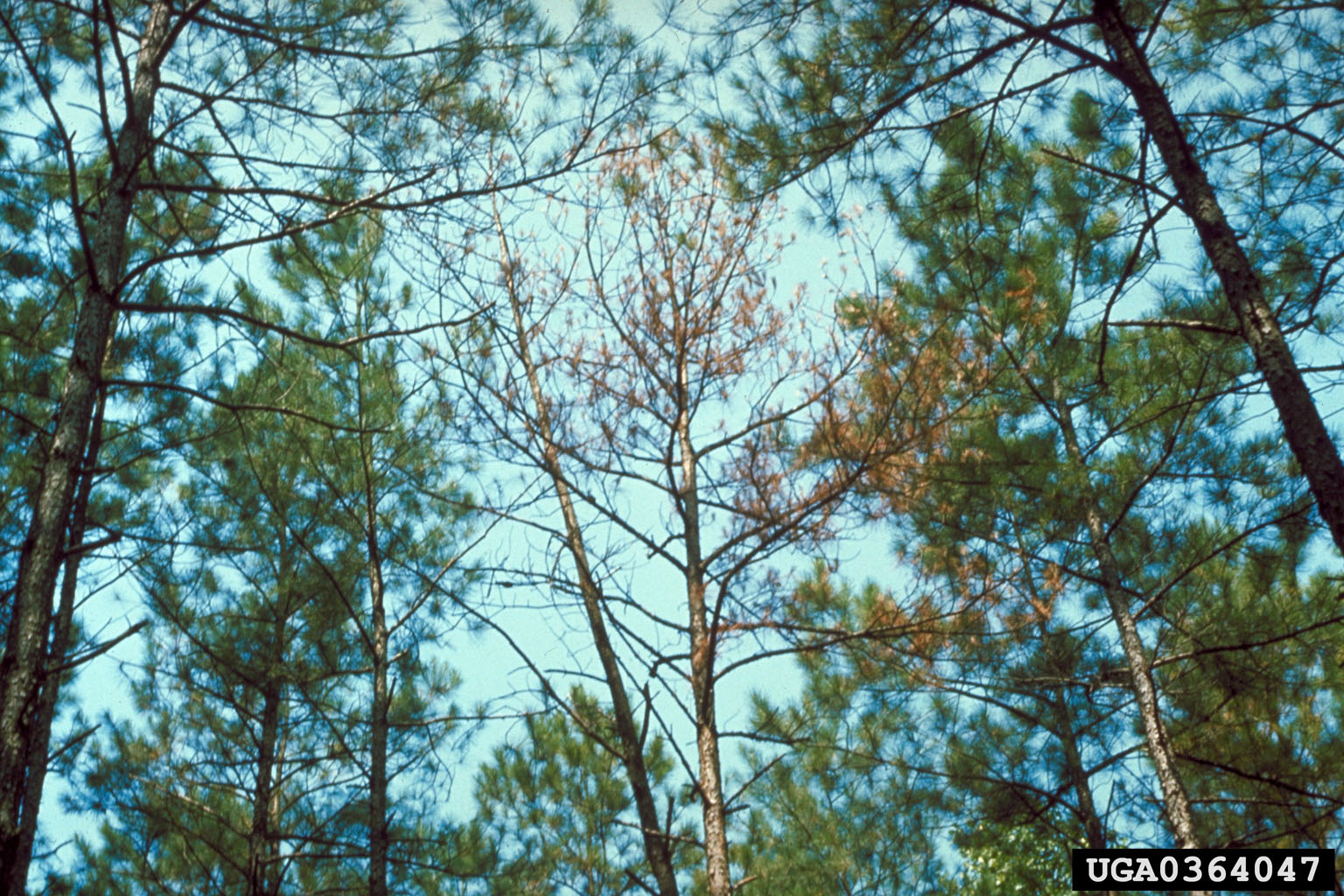
Annosum root rot occasionally becomes a problem on pines in Arkansas. It is most common on well-drained sandy soils, usually those consisting of at least 70% sand, but is not a great risk on most other sites. Annosum root rot infects trees when spores of the fungus Heterobasion annosum land on wounds of damaged trees or on freshly cut stumps after a pine stand has been thinned.
The fungus spreads from the wound or cut stump down into the root system of the tree/stump. The fungus then spreads from tree to tree through root grafts. Tree mortality due to annosum root rot occurs between two and seven years after the thinning
On soils prone to annosum infection, take steps to reduce the probability of infection. First, stands on sites prone to annosum root rot should be thinned during the summer. Annosum fungal spores do not survive well when average daily temperatures are above 70° F. Be aware that summer thinning may increase the chance of a bark beetle infestation. Call your county Arkansas Forestry Commission office and ask about bark beetle problems in your area.

Second, take great care to avoid damage to residual pines on sites with a high annosum root rot hazard. Discuss the situation with the timber buyer and logging crew foreman to make sure they understand the situation. Monitor the thinning to assure that residual trees are not being damaged.

Third, within 24 hours of a tree being cut, sprinkle at least 1/8th inch of borax on the top of the stump. The borax will prevent the spores from developing on the stump.
At planting time, one can reduce the risk of annosum root rot by planting trees at a wider spacing on sites prone to the disease. The wider spacing will delay the first thinning, reduce the number of times the stand is thinned during the rotation, and thus reduce the opportunity for annosum infection.
A wider spacing may reduce income from the stand, so discuss your decision with a local registered forester before you commit to greater tree spacing.
Loblolly pine decline (LPD) is a poorly understood syndrome resulting from the unfortunate convergence of pest problems, disease problems, and poor site conditions. LPD typically occurs in older stands of loblolly pine on sites that place the trees under stress. The stress may be result from drought on south or southwest aspects and upper slope positions, drought stress from excessively well drained soils, and drought stress from shallow hard pans that limit rooting depth. The drought stress weakens the tree making it more susceptible to attack by root weevils and bark beetles which carry Leptographium fungi that infect and destroy the root system of the tree. Weevils and beetles can also infect loblolly pine root systems damaged by severe wind whipping during storms, excessive prescribed burning, and logging activity. Trees weakened by LPD are more susceptible to attacks from bark beetles.

Symptoms of LPD often resemble those of littleleaf disease of shortleaf pine. Trees with thinning crowns and short yellowing needles scattered through a stand typically are the first symptoms of LPD observed. Trees showing LPD symptoms usually die within two to three years. There is no treatment that will cure LPD. If your loblolly pine stand shows symptoms of LPD, don’t assume the stand is lost. If less than 10% of the trees show LPD symptoms, leave the stand alone but monitor it for further decline. Remove the declining trees at the next thinning. If more than 30% of the trees show symptoms, it is time to harvest the stand and replant with a less susceptible tree or convert to an alternate land use.
Landowners can take some actions to reduce the occurrence of LPD. The only guaranteed cure for LPD is conversion to a crop other than loblolly pine. If you plan to keep the site in loblolly pine, design a management plan to minimize drought stress. Keep basal areas lower than recommended for the site. Don’t conduct prescribed burns or thin at-risk stands during drought years. If you are preparing to plant a site prone to LPD, consider planting shortleaf pine, which is less susceptible to LPD.
Littleleaf disease (LLD) is primarily a disease of shortleaf pine although it can attack loblolly pine, as well. LLD is caused by a root infection of the fungus Phytophthora cinnamomi which is a common soil fungus throughout much of the world.

Initial LLD symptoms include death of root tips and root hairs. These symptoms are out of sight in the soil so they are often overlooked. Visible symptoms of LLD include atypically short pale green needles, thin crowns, reduced twig growth, and proliferation of small cones. Infected trees often occur in groups and usually die within one to six years. Symptoms typically appear on sites with low fertility and high soil moisture. The low fertility stresses trees and prevents them from rapidly growing new roots. The high soil moisture creates an environment well suited to the Phytophthora fungus. The infertility slows root growth to such a degree that the tree cannot replace roots lost to the fungus.
Since Phytophthora fungi are found in all soils, eradication is not an option. Maintaining a healthy forest can reduce the incidence of LLD. Take steps to protect root systems when thinning stands of shortleaf pine on risky sites. At the time of reforestation, plant trees such as loblolly pine that are less susceptible to LLD. On some sites a nitrogen fertilizer may help reduce the risk of LLD.

Root rots are common among forest trees but because they remain unseen below the ground, they go unnoticed. The most common are mushroom root rot (Amalleria mellea), white root rot (Corticium galactinum), and Texas root rot (Phymatotrichopsis omnivorum). Root rots are present in the soil from previous infestations and wait on susceptible hosts. Stressed trees are more susceptible. Old cotton fields may house Texas root rot and old orchards or nurseries may have white root rot. There are no cures for infected trees and new tree planting must be delayed by 2 – 4 years until the substrate reservoir has completely decayed. It is dangerous to ignore root rot symptoms in urban trees.
This web page by University of Massachusetts Amherst, Root and Butt Rot Pathogens of Oaks, provides more information about root rots. This publication, Shoestring Root Rot - A Cause of Tree and Shrub Decline, from the University of Kentucky also provides more information about root rots.